You are looking for information, articles, knowledge about the topic nail salons open on sunday near me tris hcl ph 8.0 on Google, you do not find the information you need! Here are the best content compiled and compiled by the https://chewathai27.com/to team, along with other related topics such as: tris hcl ph 8.0
Table of Contents
How To Make 1 M Tris-HCl pH 8.0 Buffer
- Article author: toptipbio.com
- Reviews from users: 1242
Ratings
- Top rated: 4.1
- Lowest rated: 1
- Summary of article content: Articles about How To Make 1 M Tris-HCl pH 8.0 Buffer The role of Tris-HCl in molecular biology is to control the acity and osmolarity of a solution. Due to its properties, Tris-HCl is often a component of lysis … …
- Most searched keywords: Whether you are looking for How To Make 1 M Tris-HCl pH 8.0 Buffer The role of Tris-HCl in molecular biology is to control the acity and osmolarity of a solution. Due to its properties, Tris-HCl is often a component of lysis … Tris-HCl pH 8.0 is a buffering solution used to control the acidity and osmolarity of a reaction. Learn how to prepare the solution using the recipe.
- Table of Contents:
About Tris-HCl buffer solution
Download the recipe as a PDF
1 M Tris-HCl recipe
How to make 1 M Tris-HCl pH 80
Storage of 1 M Tris-HCl pH 80 solution
Safety

UltraPure™ 1M Tris-HCI, pH 8.0
- Article author: www.thermofisher.com
- Reviews from users: 1986
Ratings
- Top rated: 3.9
- Lowest rated: 1
- Summary of article content: Articles about UltraPure™ 1M Tris-HCI, pH 8.0 UltraPure™ 1 M Tris-HCl Buffers are pre-mixed, pH-adjusted, sterile-filtered solutions. Prepared as 1 M concentrates, these buffers can be diluted to the … …
- Most searched keywords: Whether you are looking for UltraPure™ 1M Tris-HCI, pH 8.0 UltraPure™ 1 M Tris-HCl Buffers are pre-mixed, pH-adjusted, sterile-filtered solutions. Prepared as 1 M concentrates, these buffers can be diluted to the … UltraPure 1 M Tris-HCl Buffers are pre-mixed, pH-adjusted, sterile-filtered solutions. Prepared as 1 M concentrates, these buffers can be diluted to the desired concentration and used in molecular biology or general biochemistry applications.<
- Table of Contents:

Tris hydrochloride solution, pH 8.0 | CAS 1185-53-1 (solid) | SCBT – Santa Cruz Biotechnology
- Article author: www.scbt.com
- Reviews from users: 9879
Ratings
- Top rated: 4.1
- Lowest rated: 1
- Summary of article content: Articles about Tris hydrochloride solution, pH 8.0 | CAS 1185-53-1 (solid) | SCBT – Santa Cruz Biotechnology pH 8.0, 1 M. Tris hydrochlore has been used as a component of extraction buffer e.g. phenol extraction of DNA or RNA; and as a component … …
- Most searched keywords: Whether you are looking for Tris hydrochloride solution, pH 8.0 | CAS 1185-53-1 (solid) | SCBT – Santa Cruz Biotechnology pH 8.0, 1 M. Tris hydrochlore has been used as a component of extraction buffer e.g. phenol extraction of DNA or RNA; and as a component … Buy Tris hydrochloride solution, pH 8.0 (CAS 1185-53-1 (solid)), non-sterile; 0.2 μm filtered, 1M stock solution used to make buffers, from Santa Cruz.[Tris HCl, NH2C CH2OH 3 HCl]
- Table of Contents:
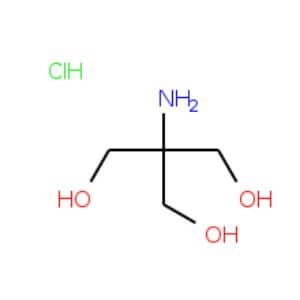
Tris-HCl
- Article author: cshprotocols.cshlp.org
- Reviews from users: 46273
Ratings
- Top rated: 4.0
- Lowest rated: 1
- Summary of article content: Articles about Tris-HCl For a 1 M solution, dissolve 121.1 g of Tris base in 800 ml of H2O. Adjust the pH to the desired value by adding concentrated HCl. … Allow the solution to cool … …
- Most searched keywords: Whether you are looking for Tris-HCl For a 1 M solution, dissolve 121.1 g of Tris base in 800 ml of H2O. Adjust the pH to the desired value by adding concentrated HCl. … Allow the solution to cool …
- Table of Contents:

Tris-HCl 1 M, pH 8.0 | Teknova
- Article author: www.teknova.com
- Reviews from users: 3161
Ratings
- Top rated: 4.9
- Lowest rated: 1
- Summary of article content: Articles about Tris-HCl 1 M, pH 8.0 | Teknova Tris-HCl 1 M, pH 8.0. Tris buffer stock for molecular biology, protein chemistry, and biochemical applications. Formulation. 1 … …
- Most searched keywords: Whether you are looking for Tris-HCl 1 M, pH 8.0 | Teknova Tris-HCl 1 M, pH 8.0. Tris buffer stock for molecular biology, protein chemistry, and biochemical applications. Formulation. 1 … Outsource your microbiology media and biological buffers and solutions preparation. Standard and custom formulations for lab and process scales.1M Tris-HCl,pH 8.0 Solution,T1080,tris ph 8.0
- Table of Contents:
Formulation
SDS
Certificate of Analysis
Additional Info

Tris-HCl Buffer (1 M, pH 8.0) | Boston BioProducts
- Article author: bostonbioproducts.com
- Reviews from users: 35275
Ratings
- Top rated: 3.6
- Lowest rated: 1
- Summary of article content: Articles about Tris-HCl Buffer (1 M, pH 8.0) | Boston BioProducts Tris-HCl Buffer (1 M, pH 8.0) – #BBT-80 · Product Description · Related Products. …
- Most searched keywords: Whether you are looking for Tris-HCl Buffer (1 M, pH 8.0) | Boston BioProducts Tris-HCl Buffer (1 M, pH 8.0) – #BBT-80 · Product Description · Related Products. Tris-HCl Buffer (1 M, pH 8.0)
- Table of Contents:

See more articles in the same category here: Chewathai27.com/to/blog.
How To Make 1 M Tris-HCl pH 8.0 Buffer
About Tris-HCl buffer solution
Tris(hydroxymethyl)aminomethane hydrochloride (Tris-HCl) is a commonly used buffering solution. The role of Tris-HCl in molecular biology is to control the acidity and osmolarity of a solution. Due to its properties, Tris-HCl is often a component of lysis buffers. The pH of the solution is most commonly made between 7.0 and 8.0.
Download the recipe as a PDF
To download the 1 M Tris-HCl recipe as a PDF then click here.
1 M Tris-HCl recipe
The recipe below is used to prepare a 100 mL 1 M Tris-HCl solution at pH 8.0. It can, however, be tweaked to make the same solution at the desired pH.
Reagent Weight / Volume Final concentration Tris base 12.11 grams 1 M Distilled H 2 O Up to 100 mL
How to make 1 M Tris-HCl pH 8.0
Weigh out 12.11 g Tris and add to a 100 mL Duran bottle. Measure out 80 mL of distilled water and add to the Duran bottle. Add a magnetic flea and place on a magnetic stirring plate to mix the solution. Add a pH meter into the solution to observe the pH. Slowly add concentrated hydrochloric acid (HCl) solution using a Pasteur pipette to reduce the pH to 8.0, or another desired pH. Be careful not to add too much at a time, since the pH will change rapidly. Once the desired pH has been reached, top up the solution to 100 mL using distilled water. To sterilise, autoclave the solution on a liquid cycle (20 min at 15 psi).
Storage of 1 M Tris-HCl pH 8.0 solution
Store 1 M Tris-HCl pH 8.0 solutions at room temperature (+15oC – +25oC).
Safety
Tris base is not classed as a hazardous substance, however, be careful when using HCl during the pH adjustment step. Concentrated HCl solution is an acid as is very corrosive. Always read the material safety data sheet before using it.
Tris hydrochloride solution, pH 8.0 | CAS 1185-53-1 (solid)
pH 8.0, 1 M. Tris hydrochloride has been used as a component of extraction buffer e.g. phenol extraction of DNA or RNA; and as a component of separating and stacking gels in the characterization of in vitro translation products by SDS-PAGE. The pH values of Tris buffer solutions are temperature and concentration-dependent. Between 5 °C and 25 °C, the pH value increases an average of 0.03 pH units for each °C decrease in temperature. As the buffer temperature increases from 25 °C to 37 °C, the pH value decreases an average of 0.025 pH units per °C. Increasing the concentration of Tris from 0.05 M to 0.5M will increase the pH value by about 0.05 units. Decreasing the concentration from 0.05 M to 0.005 M will decrease the pH value by about 0.05 units.
Tris-HCl
For a 1 M solution, dissolve 121.1 g of Tris base in 800 ml of H 2 O. Adjust the pH to the desired value by adding concentrated HCl.
pH HCl 7.4 70 ml 7.6 60 ml 8.0 42 ml
Allow the solution to cool to room temperature before making final adjustments to the pH. Adjust the volume of the solution to 1 liter with H 2 O. Dispense into aliquots and sterilize by autoclaving.
If the 1 M solution has a yellow color, discard it and obtain Tris of better quality. The pH of Tris solutions is temperature-dependent and decreases approx. 0.03 pH units for each 1°C increase in temperature. For example, a 0.05 M solution has pH values of 9.5, 8.9, and 8.6 at 5′C, 25′C, and 37°C, respectively.
So you have finished reading the tris hcl ph 8.0 topic article, if you find this article useful, please share it. Thank you very much. See more:

